The Gut–Liver–Immunity–Kidney Axis: How Gut Health Influences Kidney Function
Written by Prem Nand, Integrative Clinical Dietitian-Nutritionist, NZRD
Introduction: Health Is a Circulating Story
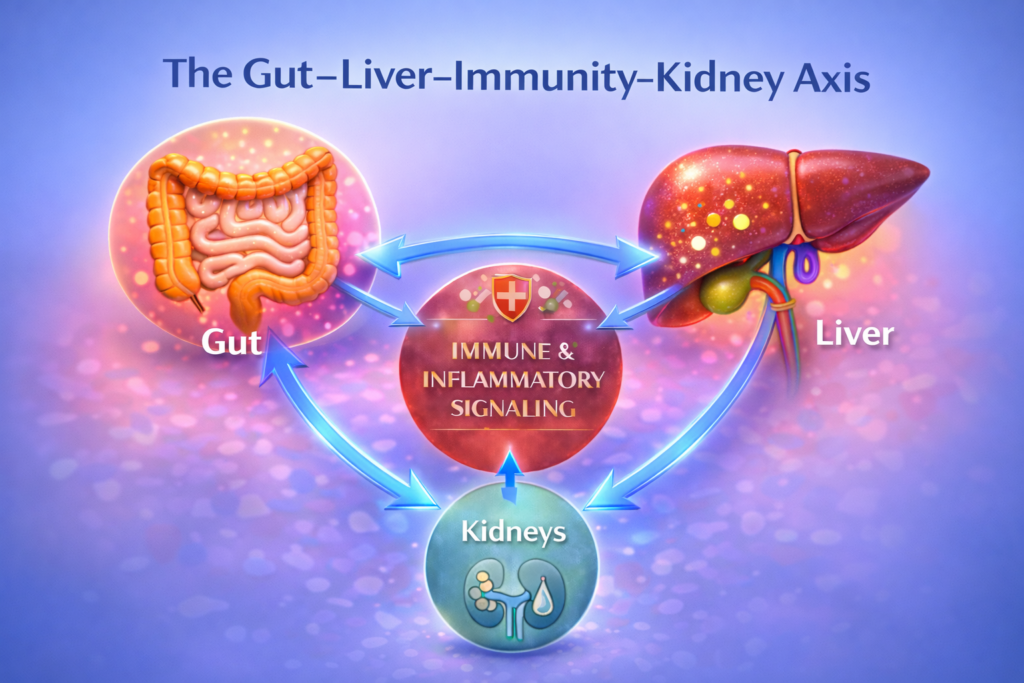
The gut–liver–kidney axis describes the continuous biological relationship between digestion, immunity, liver processing, and kidney filtration. These systems are not separate compartments. They communicate constantly through circulation, immune signalling, metabolic by-products, and inflammatory mediators.
In chronic illness, this interconnectedness becomes more visible.
Consider a woman in her mid-40s with lifelong bowel dysfunction who later developed kidney disease due to focal segmental glomerulosclerosis (FSGS). Over time she required a kidney transplant. Years later, she presents with persistent gut symptoms, confirmed SIBO-hydrogen and intestinal methanogen overgrowth (IMO), deranged liver enzymes, and ongoing fatigue.
This story is not about one organ causing another to fail. It is about how biological stress can circulate — literally — through the body over time.
Understanding the gut–liver–kidney axis helps us see that circulation is not just about blood flow. It is about information flow.
The Gut: Where the Story Begins
The gut is more than a digestive tube. It is an immune organ, a metabolic processor, and a microbial ecosystem. Trillions of microbes live in the gastrointestinal tract, producing short-chain fatty acids, modifying bile acids, synthesising vitamins, and interacting directly with immune cells (Kho & Lal, 2018).
When balanced, this ecosystem supports:
• Barrier integrity
• Immune tolerance
• Nutrient absorption
• Metabolic regulation
When disrupted — a state known as gut dysbiosis — digestive symptoms often appear first. But the consequences may extend further.
In SIBO and IMO, bacteria and methane-producing organisms colonise regions of the small intestine where microbial density is normally low. This can alter motility, increase fermentation, and irritate the gut lining (Pimentel et al., 2020). Gas production, bloating, slowed transit, and malabsorption are common features.
More subtly, dysbiosis may affect the integrity of the intestinal barrier.
The Gut Barrier, Immune Signaling
The gut lining functions as a selective filter. It allows nutrients into circulation while keeping microbes and bacterial fragments contained within the intestinal lumen.
When barrier integrity weakens, small amounts of microbial components such as lipopolysaccharide (LPS) may enter circulation. This does not mean infection.
It means the immune system detects signals that it interprets as potential threat.
Even low-grade microbial translocation can sustain inflammatory signaling through innate immune pathways (Tripathi et al., 2018). This process may be silent — without fever or acute illness — yet still biologically meaningful over time.
Once these signals enter circulation, they do not remain in the gut.
They travel.
From Gut to Liver: The Portal Connection
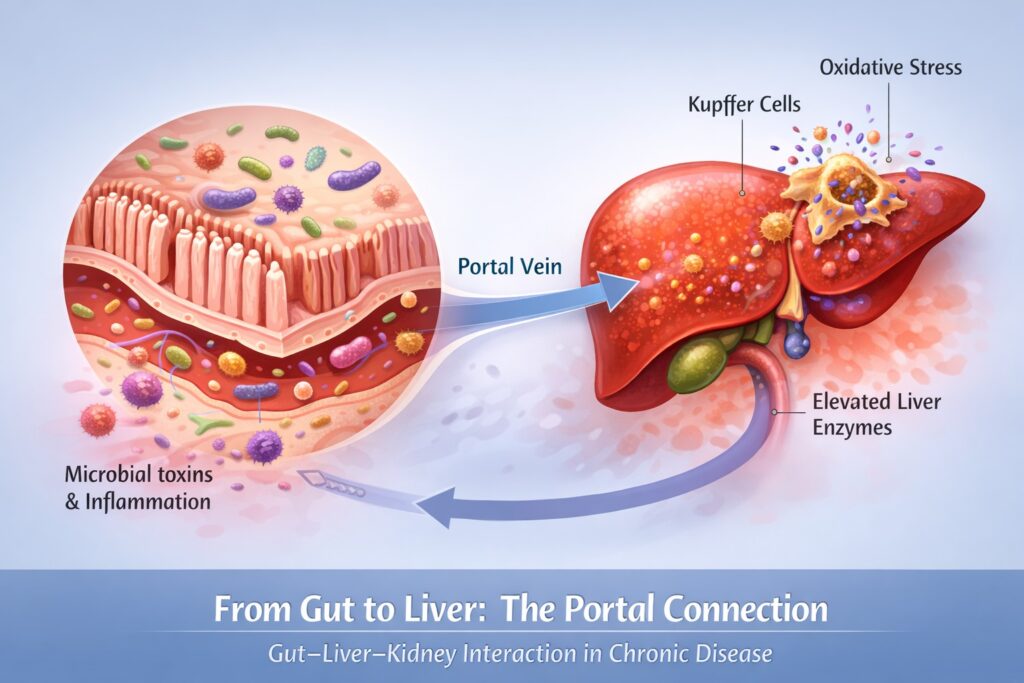
Everything absorbed through the intestines travels first to the liver through the portal vein. This anatomical arrangement ensures that the liver acts as a metabolic checkpoint.
Under healthy conditions, the liver processes nutrients, detoxifies compounds, and maintains immune tolerance. But when microbial metabolites and inflammatory signals increase, hepatic workload rises.
Research describing the gut–liver axis demonstrates how microbial imbalance and increased permeability can influence hepatic immune activation and metabolic regulation (Albillos et al., 2020; Tripathi et al., 2018).
Kupffer cells — the liver’s resident immune cells — respond to circulating microbial fragments. Cytokine production may increase. Oxidative stress may rise. In some individuals, liver enzymes become elevated.
Importantly, abnormal liver function tests in complex chronic illness often reflect inflammatory stress rather than structural liver damage.
The liver, however, does not hold onto what it processes.
It passes it forward.
From Liver to Kidney: Systemic Circulation and Filtration
After hepatic processing, metabolites and inflammatory mediators enter systemic circulation. At this stage, the kidneys become central.
The kidneys filter blood continuously. They remove waste products, regulate electrolytes, and maintain fluid balance. When kidney reserve is reduced — as in FSGS — the capacity to buffer metabolic and inflammatory load declines.
Over time, chronic kidney disease alters internal metabolism in ways that also affect the gut environment. Uremic toxins accumulate. Intestinal pH changes. Microbial composition shifts.
This is where the relationship becomes clearly bidirectional.
The Gut–Kidney Axis: A Two-Way Relationship
A landmark review in Kidney International describes how chronic kidney disease is associated with gut microbial changes, impaired barrier function, and abnormal immune activation (Anders, Andersen, & Stecher, 2013).
As kidney function declines, uremia alters the intestinal environment. This can promote dysbiosis and weaken the gut barrier. Microbial products may translocate more easily, sustaining immune activation and systemic inflammation.
At the same time, certain toxins produced by gut microbes — including indoxyl sulfate and p-cresyl sulfate — accumulate in kidney disease and contribute to oxidative stress and renal fibrosis (Ramezani & Raj, 2014).
Thus, the gut influences the kidney, and kidney impairment reshapes the gut.
This circular relationship defines the gut–liver–kidney axis.
The Immune System: The Integrating Network
Running through this entire axis is the immune system.
It senses microbial signals in the gut.
It activates within the liver.
It influences inflammatory pathways in the kidneys.
Pattern-recognition receptors respond to microbial components, even at low concentrations (Tripathi et al., 2018). Over time, repeated exposure may contribute to immune priming — a state in which inflammatory responses become amplified.
Chronic low-grade inflammation has been described in kidney disease, liver dysfunction, and metabolic disorders. It can impair tissue repair and promote fibrosis.
Inflammation also affects mitochondria — the energy-producing structures inside cells. Mitochondrial dysfunction has been documented in chronic kidney disease and contributes to fatigue and reduced physiological resilience (Forbes & Thorburn, 2018).
Thus, immune signaling is not an isolated event. It is a networked process linking organs through circulation.
Transplant Vulnerability and Systemic Balance
In kidney transplant recipients, immune regulation becomes even more delicate.
Immunosuppressive medications may alter the microbiome. Barrier integrity may fluctuate. Subtle inflammatory signals may carry greater consequence.
Emerging research suggests that gut microbiota composition may influence immune signaling relevant to transplant outcomes (Lee et al., 2020). This does not imply that gut health determines graft success, but it reinforces the importance of systemic balance.
In individuals with reduced renal reserve or transplanted kidneys, cumulative inflammatory load may matter more.
Seeing the Feedback Loop Clearly
At this stage, the physiological flow becomes visible:
1. Gut imbalance alters barrier integrity.
2. Microbial signals reach the liver.
3. Hepatic processing influences systemic inflammatory tone.
4. The kidneys filter circulating metabolites under varying reserve.
5. Reduced renal clearance reshapes the gut environment.
6. The cycle continues.
This is not a straight line. It is a loop.
The gut–liver–kidney axis describes this circulating feedback system — one that may amplify stress when multiple organs are already vulnerable.
What This Means for Chronic Illness
Understanding the gut–liver–kidney axis helps explain why symptoms overlap in chronic disease:
• Digestive instability
• Fatigue
• Fluctuating liver enzymes
• Inflammatory markers
• Renal vulnerability
It also explains why stabilising one system may reduce pressure on others.
This does not mean reversing structural kidney disease through dietary change alone. FSGS and transplant management require specialised medical oversight.
However, reducing unnecessary inflammatory burden — particularly from the gut — may support systemic resilience alongside conventional care.
Conclusion: Seeing the Body as a Connected System
The gut–liver–kidney axis offers a practical, evidence-based way of understanding chronic illness as a whole-body experience, rather than a collection of isolated conditions. For people living with long-standing digestive issues, immune stress, liver dysfunction, and kidney vulnerability, this lens helps make sense of why symptoms are often interconnected and persistent.
By acknowledging how these systems influence one another, care can become more supportive, integrated, and realistic — focusing less on quick fixes and more on reducing cumulative strain on the body. While complex chronic disease cannot be reversed by any single strategy, even modest improvements in gut balance or inflammatory load may help support stability and resilience alongside ongoing medical care.
References
Albillos, A., de Gottardi, A., & Rescigno, M. (2020). The gut–liver axis in liver disease. Journal of Hepatology, 72(3), 558–577.
Anders, H.-J., Andersen, K., & Stecher, B. (2013). The intestinal microbiota, a leaky gut, and abnormal immunity in kidney disease. Kidney International, 83(6), 1010–1016.
Forbes, J. M., & Thorburn, D. R. (2018). Mitochondrial dysfunction in diabetic kidney disease. Nature Reviews Nephrology, 14(5), 291–312.
Kho, Z. Y., & Lal, S. K. (2018). The human gut microbiome. Frontiers in Microbiology, 9, 1835.
Lee, J. R., Muthukumar, T., Dadhania, D., et al. (2020). Gut microbiota and transplant outcomes. Transplantation, 104(4), 718–726.
Pimentel, M., Saad, R. J., Long, M. D., & Rao, S. S. (2020). ACG guideline: Small intestinal bacterial overgrowth. American Journal of Gastroenterology, 115(2), 165–178.
Ramezani, A., & Raj, D. S. (2014). The gut microbiome and kidney disease. Journal of the American Society of Nephrology, 25(4), 657–670.
Tripathi, A., Debelius, J., Brenner, D. A., et al. (2018). The gut–liver axis. Nature Reviews Gastroenterology & Hepatology, 15(7), 397–411.
Vaziri, N. D. (2012). CKD alters intestinal flora. American Journal of Nephrology, 36(5), 438–443.